
Citric Acid, the Krebs cycle intermediate, is enzymatically dehydrated in the mitochondria of cells to generate (Z) aconitic acid.
In addition, the non-enzymatic dehydration route to generate (E) aconitic using concentrated sulfuric acid sulfuric acid is well known. A similar route using a
catalytic amount of the less oxidizing phosphoric acid at 120°-150°C has also been used to dehydrate citric acid
(Umbdenstock & Bruins, Ind. Eng. Chem 1945, 37(10) p 963-967). Aconitic acid along with its decomposition products, itaconic and citraconic acids, has also been
produced by the decomposition of citric acid, in the melt phase
In another method patented by Smeets, US3586717, dry calcium aconitate was produced from dry calcium citrate during a 6 hour period at temperatures of
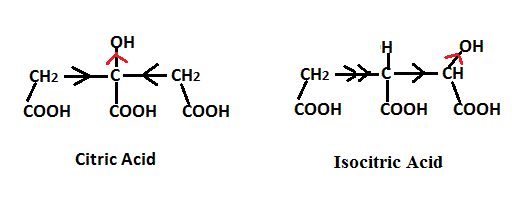
Citric Acid which has the structure shown below (as the sawhorse projection on the left and the Newman projection on the right).
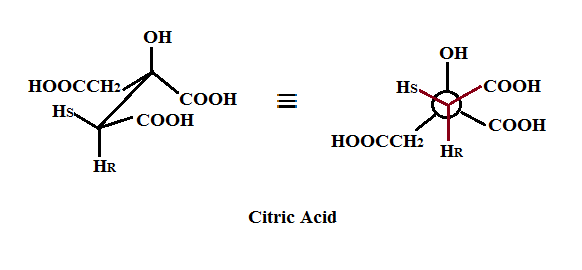
can be enzymatically converted via Krebs cycle conditions to (Z) aconitic acid. The dehydration step involves the removal of the HR from the R face of citric acid via a concerted E2 mechanism.
Is it possible to convert citric to aconitic at near neutral non-enzymatic conditions? We have shown that the isocitrates can be converted to the aconitic acids and then to citric acid under basic conditions. In addition, strong acid conditions favor the dehydration of citric by protonation of the hydroxyl group making it a better leaving group. However, we would like to investigate the dehydration at near neutral/weak acid conditions (pH 4-6.5) where the reaction conditions are less severe. The dehydration step, however, would require a weakly nucleophilic step to remove either one of the diasterotopic hydrogens HR or HS.
Why either one? Since the dehydration reaction is taking place under non enzymatic Krebs cycle conditions, the reaction is non stereospecific and can lead to either proton HR or HS being abstracted (as long as either one is trans to the OH group). Since there are two enantiotopic HRs and two enantiotopic HSs, then either the R or S face of citric acid is susceptible to attack and, unlike the enzymatic Krebs reaction, would lead to a mixture of products.
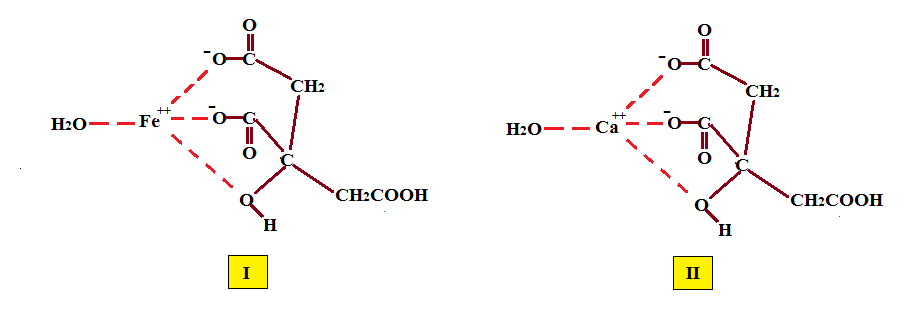
Citric acid is known to form tridentate complexes with metal ions. With Fe+2 the critical stability constant (CST) is 3.2 and its structure is (I) according to Francis & Dodge in (a) Applied & Environmental Microbiology Jan 1993 pp 109-113 and (b) aem.asm.org/content/59/1/109.full.pdf. Similarly the calcium salt (II) with a CST of 3.5 should exhibit similar properties, As discussed by Wyrzykowski et al J. Therm. Anal. Calorim. 102:149-154 in the thermodynamic interaction of alkaline earth metal ions with citric, the citrate acts as a tridentate ligand. The X-ray crystallographic pattern shows that the central carboxyl of citric is perpendicular to the carbon backbone and lies in one plane with the hydroxyl group in bidentate fashion while a terminal carboxyl groups acts as the third donor as shown in (II).
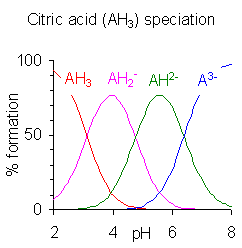
Since we are investigating the possible dehydration of citrate at weakly acid pHs, we should determine what species are present at the requisite pHs, i.e., in the range of 4-7. To do this we look at the citric acid speciation chart which shows the concentration of the four species, one of which is fully protonated, at pHs ranging from 2 to 8 (borrowed from Pedersen at Wikepidia.com). Specifically between a pH of 5-7 the AH2- species is present in about 80%. The AH1- and the AH3- are only present as the rest of the 20%. Thus if we use this graph in conjunction with calcium as the cation, then the species we are most interested in is the mono calcium citrate salt at a pH of about 5.8. We are less interested in the half calcium salt species or the fully salt form (Ca3 Citrate2).
Why the calcium salt? Calcium tends to bind adjacent carboxyl groups and hold them together on one side of the molecule as opposed to the sodium salts which would permit the carboxylate groups to rotate readily about their bonds.
Let us look at the induction effects of the various CHs of Citric vs Isocitric acids. Citric has two CH2 groups having a I+ effect on the molecule and the middle carbon hydroxyl group is electron withdrawing (I- effect). The middle carbon, therefore, acquires a δ+ charge which is transmitted through the sigma bonds to the electron donating CH2 groups on either side of the middle carbon, in effect spreading out the + charge amongst the four CHs.
Isocitrate, on the other hand, bears a secondary alcohol and only one α CH group and one β CH2 both which when combined have a lower I+ effect than the two CH2s of citrate, resulting in more of a δ+ charge on the secondary carbon. As a result the end CH2 groups of citric bearing the more δ+ charge, of which there are four, are more prone to attack by a nucleophile than the CH group of isocitrate, which requires a rather high pH to remove the α hydrogen as was shown in the generation of citric acid .
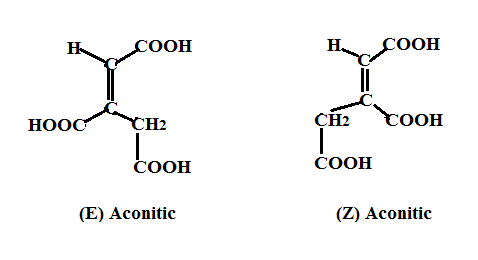
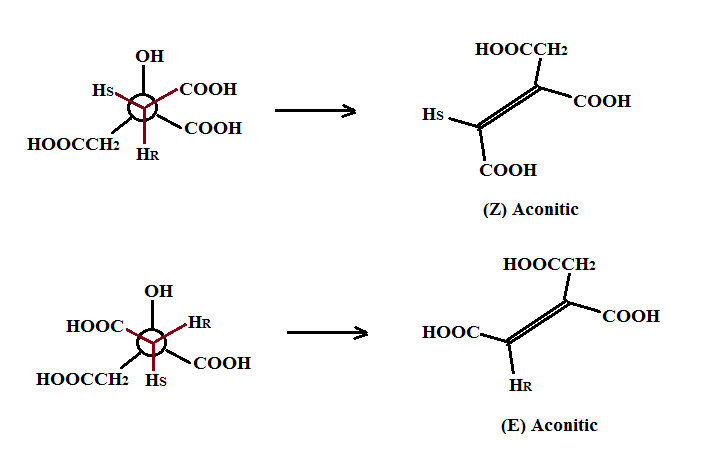
Dehydration of Citric acid by non-enzymatic mean, such as for example using conc. sulfuric acid, is no longer subjected to the stringent enzymatic conditions of the Krebs cycle. The dehydration step can lead to the removal of either of the diastereotopic four hydrogens (R or S) from either of the two α carbons. Thus removal (from the R face) of the HR leads to (Z)-aconitic while removal of the HS leads to (E)-aconitic as shown below:
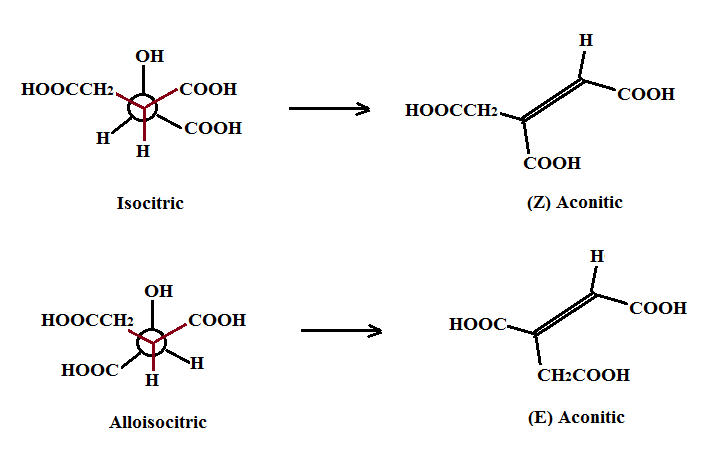
The dehydration step involving isocitric or alloisocitric via a non-enzymatic step, as well, should also produce (Z) and (E) aconitics as shown below:
According to the speciation graph above the four species in solution are AH3, AH2-, AH2- and A3-. Thus, the strongest acids in the group begin at the far left and decrease in strength as follows:
while the strongest conjugate bases begin at the far right and decrease in strength as follows:
where AH2- refers to the deprotonation of the middle carboxylate, AH2- to one of the terminal CH2 carboxylates and A3- to the second CH2 carboxylate.
Similarly with the isocitrics using the same acid/conjugate base strengths as above, the AH2- corresponds to the terminal carboxylate adjacent to the OH, the AH2- to the middle carboxyl and A3- to the terminal CH2 carboxylate. Why look at these acid/conjugate base strengths? I will show that the removal of the trans hydrogen during the dehydration step will involve the use of the strongest conjugate base at the requisite pH. Since the pKas of citric are 3.1, 4.77 and 6.39 while those of isocitric are 3.29, 4.71 and 6.40, we are looking at a pK2 of 4.7 close to the pH of 5.8 desired.
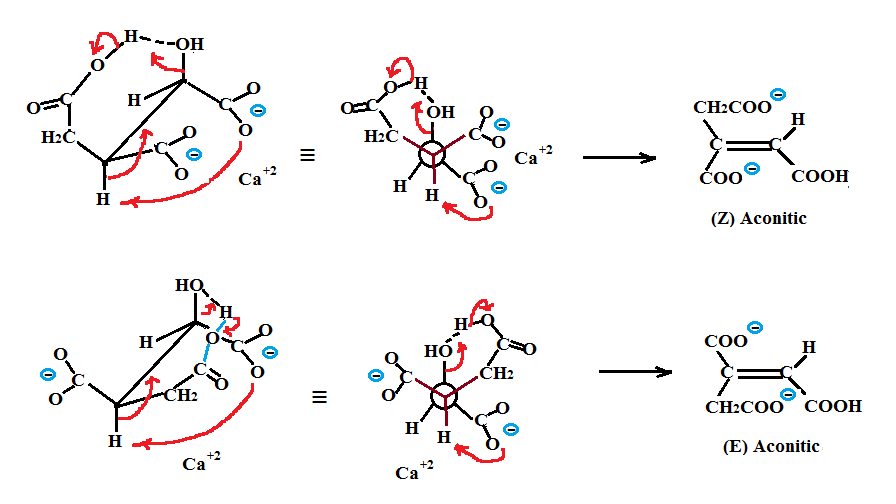
Dehydration of citric acid via non enzymatic means (we are talking high pressure, aqueous solutions and temperatures of about 200°C) would produce both (Z) and (E) aconitics as shown in the next figure. The mechanism of dehydration for the two citric salt forms is shown in both their sawhorse and Newman projections. In the figure, the calcium ions are used to hold the middle carboxylate and one of the terminal CH2 carboxylate groups together while the second carboxylic acid (the weakest acid) hydrogen bonds with the central OH group. Dehydration via deprotonation of either the HR or HS with the the second strongest conjugate base, a terminal carboxylate, followed by the concerted OH elimination results in the production of either (Z) or (E) aconitics. Such a mechanism could be interpreted as an intramolecular carboxylate assisted dehydration.
Note that both the deprotonation of the HR or HS and the hydrogen bonding of the OH occur via an intermediate with two 6 membered rings. Deprotonation via a five membered intermediate with the central carboxylate is less favorable since this would involve the weakest conjugate base.
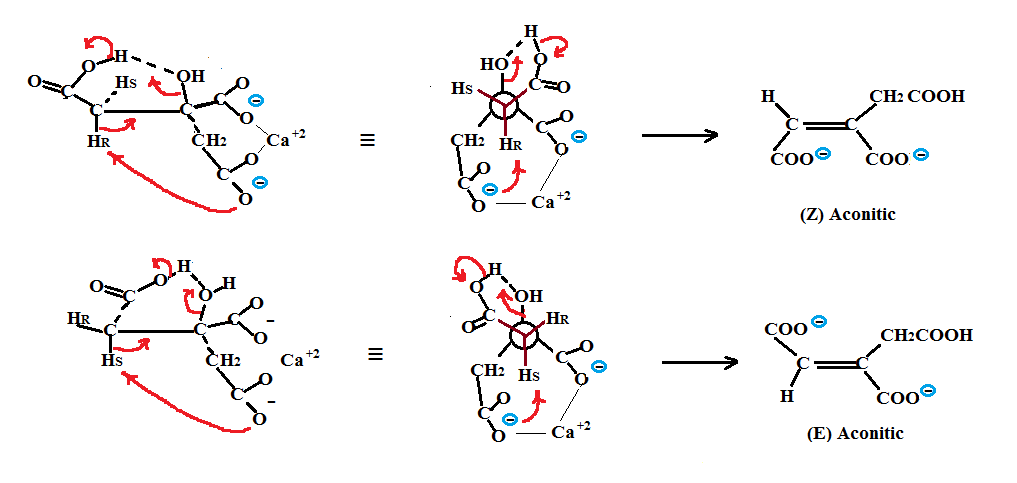
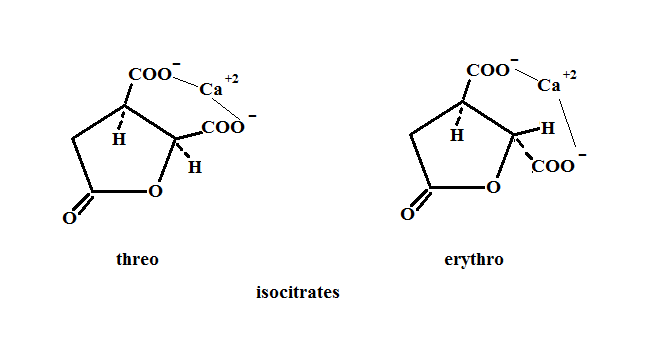
I will show that dehydration of the Isocitric acids via non enzymatic means (again high pressure, aqueous solutions and temperatures of about 200°C) to produce either (Z) and (E) aconitics is not as viable an option as it was for citric acid. The sawhorse and Newman projections of the dehydration show that a seven membered ring is used in hydrogen bonding to the OH group and that a five membered ring with the weakest conjugate base adjacent to the secondary OH group is used for deprotonation. Such an arrangement would probably require a higher energy and as I said previously would be less likely to occur. We also know that under highly basic conditions the isocitrates dehydrate to aconitic and rehydrate to citric as was shown in the paper generation of citric acid .
We must also consider the lactone forms of the monocalcium isocitate. Both these acids have their terminal CH2 carboxylates tied up as lactones and thus unavailable for hydrogen bonding. Dehydration via the intramolecular carboxylate assisted route is, therefore, more difficult. The lactones, on the other hand, can hydrolize under the reaction conditions employed. However, this produces the isocitric acids which brings us back to square one.
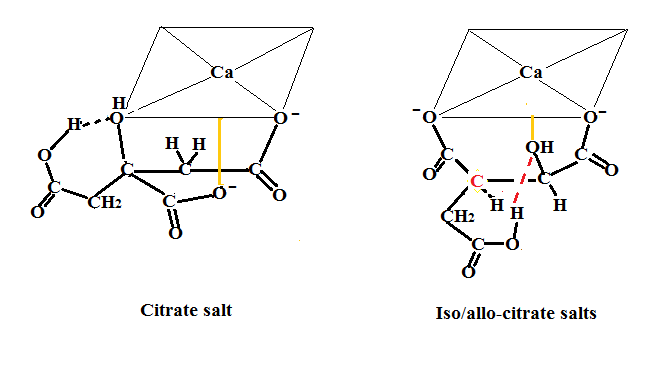
As mentioned previously citrate and isocitrate/alloisocitrate salts complex with calcium in a tridentate fashion. Below is shown how these salts chelate the calcium ion. Since this tridentate chelation would make the above figures too cumbersome to visualize this type of bonding is shown here and shoud be considered when looking at the total picture. Thus we can concentrate on just the mechanism above and keep things simple.
This figure was modified slightly from J Glusker entitled Structural Aspects of Citrate Biochemistry in Current Topics in Cellular Regulation Volume 33 P180 where the metal ion used was potassium. Note that the red carbon is used to distinguish between the two diastereomers isocitric and alloisocitric. In the left structure for citrate, the OH is over to the left while in the right structure for the isocitrates it is situated in the middle. It can be seen that in citrate, hydrogen bonding from carboxylic acid to the OH is via a six membered ring while in the isocitrates it is via a seven membered ring.
Aconitic acid walks into a bar and says to the bartender, "I'm so dehydrated gimme a double shot of water". After several shots the bartender says, "I think you've had enough. You're completely hydroxylated". The besotted drinker says back, "I think sho too but I feel like a whole new molecule, so don't citricshize me."
Go back to homepage.
Copyright © June 2015 by Eddie N Gutierrez E-mail: enaguti1949@gmail.com