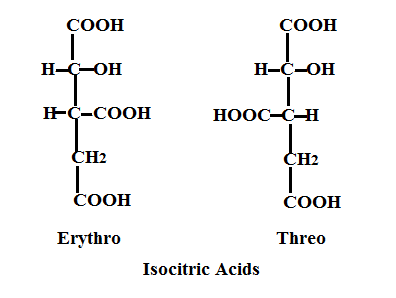

Isocitric Acid, an intermediate in the well known Krebs cycle, is normally produced under enzymatic conditions in the mitochondria of cells. However, a simple, non-enzymatic high yield route to a mixture of isocitric and alloisocitric acid was not available until 1977 when Lever Brothers, a division of Unilever, was granted a patent for the chemical production via the oxidation of compounds of 1,1,2,3 propane tetracarboxylic acid or esters.
The reactions to produce erythro and threo isocitrics employ non-enzymatic non-Krebs cycle routes and are described in US4022803 E. Gutierrez, V. Lamberti. The starting materials, 1,1,2,3,propane tetracarboxylic acid or its esters (PTCA), are halogenated (by a hapohalite: chlorine or bromine). It can also be prepared by the reaction of PTCA by H2O2/catalytic iodine while not included in the patent, is described in the isocitric /HOI reaction. Both reactions generate the isocitric mixture when the starting material is the acid at a pH of about 5. Below is the reaction of the tetracarboxylic acid (I) with hypochlorite as the oxidizing agent via an oxidative decarboxylation of the methine carbon to form the mixture of isocitric acid lactones.
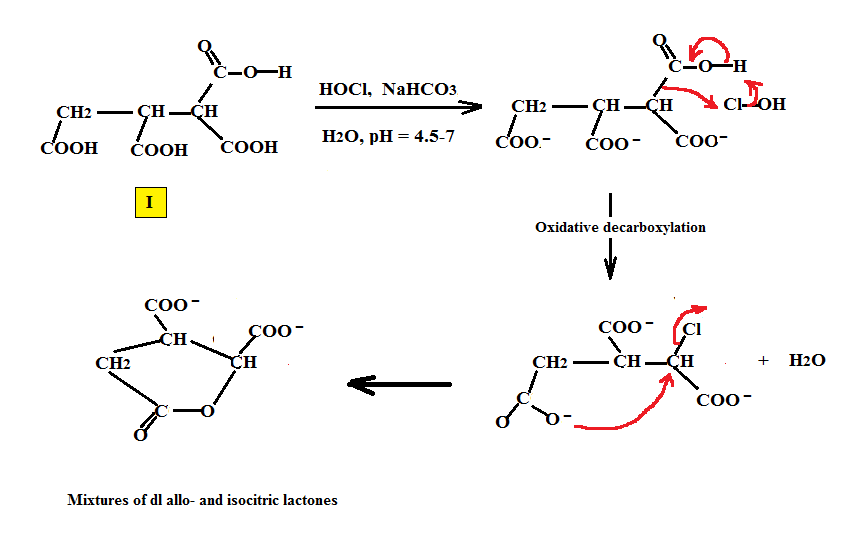
Alternatively, the decarboxylation step can occur after the halogenation step is complete. Both mechanisms are possible.
On the other hand, when the triester (II) is used (US4275007 E. Gutierrez, V. Lamberti) the chloro triester (III) as well as the four membered β-lactone (IV) are isolated with complete conversion to this lactone when pyridine is used. The presence of the two gem carboxylated ester groups in III appears to aid in closing the four membered ring. When the starting tetraester (V), (US4022803 E. Gutierrez, V. Lamberti) is employed, the isolable halo tetraester (VI), is isolated. Subsequent hydrolysis under acidic conditions of (III),(IV) and (V) results in the formation of both isocitrates.
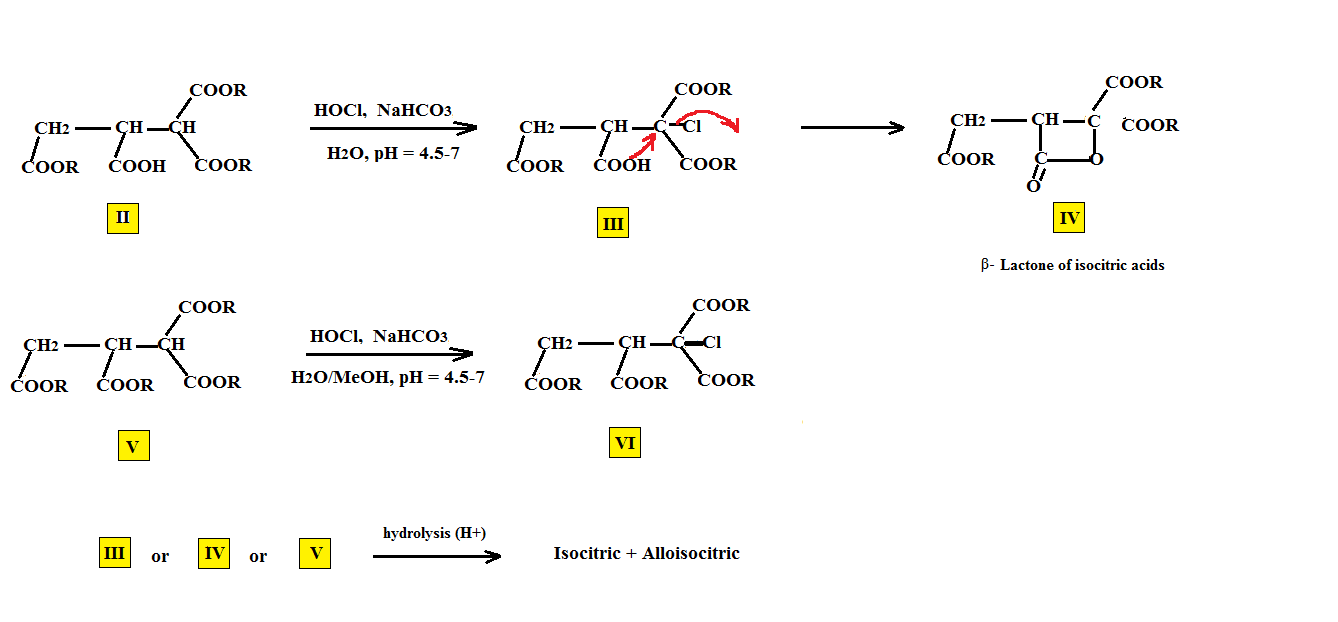
Under none of these conditions is the alternative isomer citric acid formed. We have shown that it can be prepared in Part I but only during the hydration of cis aconitic under basic conditions.
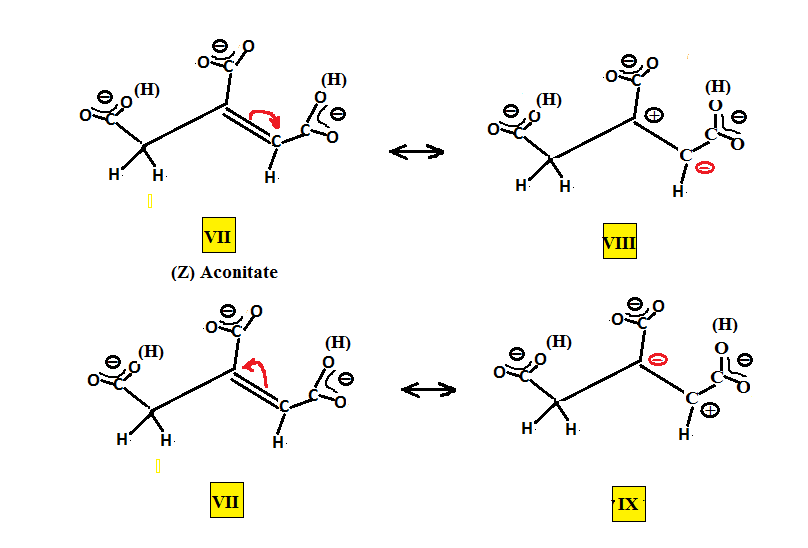
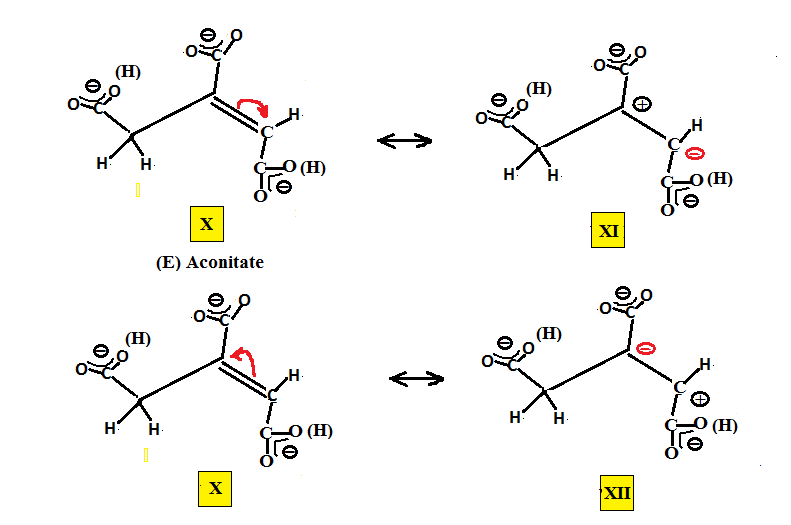
The resonance structures for cis and trans aconitic may be set up as shown below. The counter ion is left blank since both alkali metal and alkaline earth metal ions may work. The (H)s adjacent to the carboxy groups mean that either one of these is partially deprotonated. The middle carboxy being the more acidic is present as COO- but at a pH of about 4.5-5 either of the terminal COOH groups might be present as COO-. We can obtain two resonance forms for each, VIII and IX for the (E) aconitic and XI and XII for (Z) aconitic, where through resonance both the terminal and central carbons may have either both positive and negative charge for one resonance form or negative and positive charge for the other resonance form.
What these resonance forms show is that either carbon is susceptible to nucleophilic attack and what is needed is a means for distinguishing between the reactivity of both carbons.
Go back to homepage.. To go to Part III Non-Enzymatic Citric Acid Dehydration.
© April 2015 (revised 2/25/2025) by Eddie N Gutierrez E-mail: enaguti1949@gmail.com